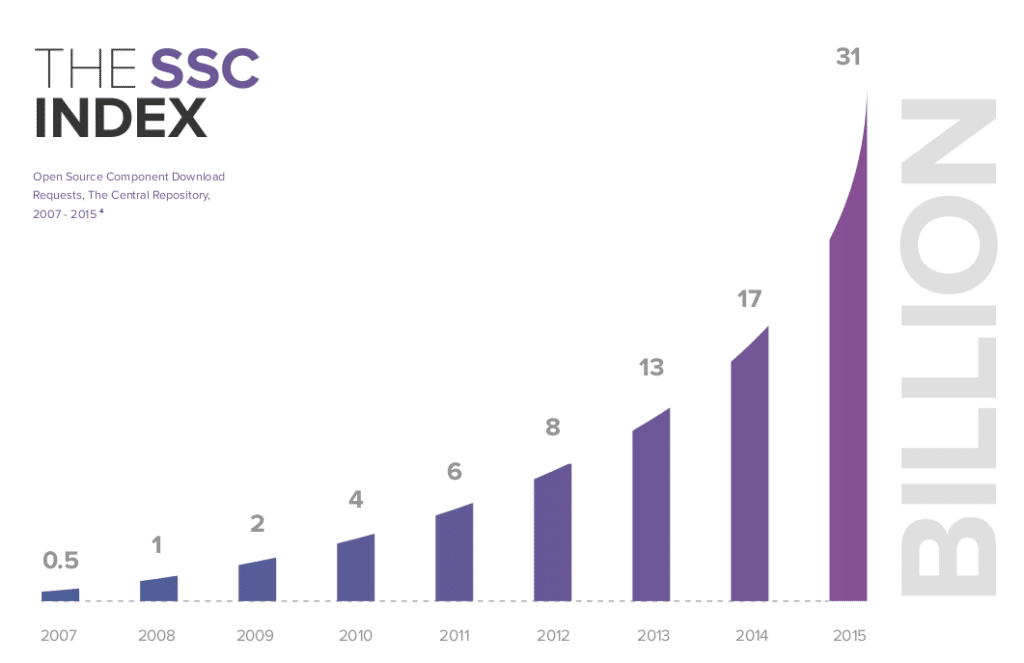
In-brief: The use of open source software is exploding, but concerns about code quality and security in the open source supply chain persist, according to a report from the firm Sonatype.
Sonatype
FDA: Medical Device Makers Urged To Secure Post Market Devices from Cyber Attack
In-brief: The U.S. Food and Drug Administration (FDA) on Friday issued new guidelines, calling on medical device makers to do a better job addressing cyber security vulnerabilities and exploits as part of their management of deployed medical devices.
Report: Fear of lawsuits chills car hack research | TheHill
In-brief: The Department of Transportation is weighing policies governing independent security researchers’ work on connected vehicles. But security industry experts worry that overreach could put a chill on independent research on connected cars.
Past and Future Risks Bedevil Security on Internet of Things
In-brief: Efforts to secure the Internet of Things will be challenged both by a backlog of old software and hardware, and by the rapid pace of technology evolution, experts warned at the recent Security of Things Forum in Cambridge, MA.