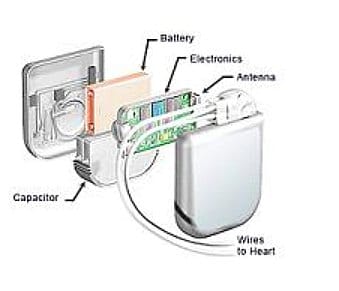
Software used to remotely program implantable cardiac devices by a number of vendors is rife with exploitable software vulnerabilities that leave the devices vulnerable to attacks and compromise, according to a report by the firm Whitescope Inc.
Tag: medical devices
Google Looks Beyond Passwords To Secure Data, Assets
In-brief: Google is pushing an approach to network security dubbed “tiered access,” demoting the trusted password, which is now just one piece of data that is needed to get access to sensitive data and resources on Google’s network.
Update: FDA says St. Jude Medical knew about Device Flaws 2 Years Before Muddy Waters Report
In-brief: In a damning report, the FDA said that St. Jude Medical* knew about serious security flaws in its implantable medical devices as early as 2014, but failed to address them with software updates or other mitigations, or by replacing those devices. (Editor’s note: updated to include a statement from Abbott and comment from Dr. Kevin Fu. – PFR April 14, 2017)
FBI Warns Medical Offices: Exposed FTP Servers are a Target
In-brief: The FBI is telling medical and dental offices to lock down anyFile Transfer Protocol (FTP) servers in their environments, warning that cyber criminals are searching for exposed FTP servers as a pathway to sensitive networks and protected health information (PHI).
Silent Epidemic: Data Theft has become a Public Health Crisis | Digital Guardian
In-brief: One in four Americans was the victim of data theft, but policy makers can’t find the spirit to act. What if I said that there was a disease that affected one in four adults in the U.S.? This disease caused pain and hardship and the costs of curing it were considerable – ranging to thousands of dollars per patient? And, again: one in four people contacted this disease – 25% of the adult population?Most epidemiologists would consider a disease that widely spread to be an “epidemic.” After all, the CDC considers an influenza outbreak to be an “epidemic” when around 7% of morbidity (deaths) in a given observation period are due to the flu. Here in the U.S., however, there’s a long-standing affliction bearing down on a quarter of the population, but nary a mention of the words “epidemic.” In fact, officials who monitor this disease are loath to […]