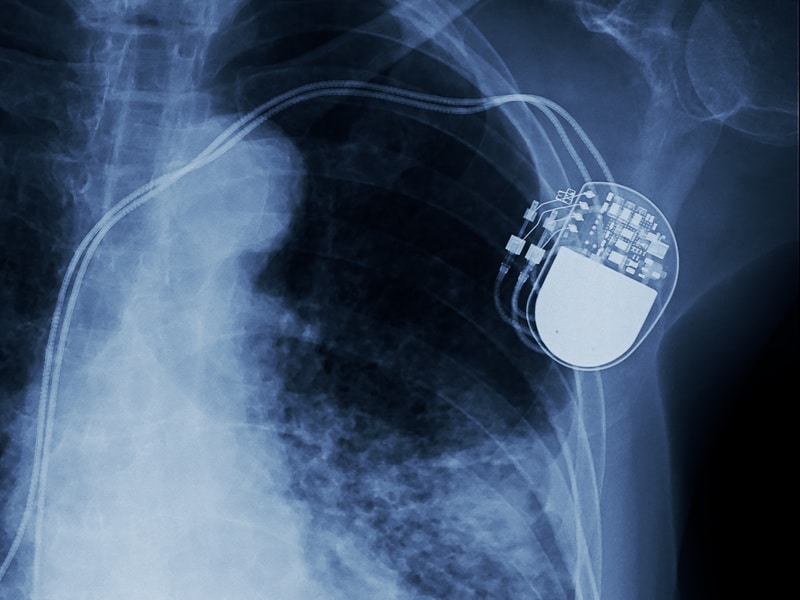
In this Spotlight Podcast, sponsored by Synopsys: In the wake of a presentation at Black Hat about security flaws in implantable pace maker devices, Synopsys Principal Consultant Dan Lyon joins us to talk about why medical device makers struggle to make their connected medical devices more secure. Dan and I discuss some of the flaws in the approach that medical device makers take to security, and how manufacturers can take a page out of their own book: applying the same standards to cyber security as they do to – say- device safety.
Tag: medical devices
From Mainframes to Connected Cars: How Software drives the Automotive Industry
Automakers must pay as much attention to the integrity and security of the software running modern vehicles as they pay to areas such as metallurgy, impact protection, seat belts, and materials science argues Gary Mcgraw, the Vice President of Security Technology at the firm Synopsis.
Spotlight Podcast: CSS on why Crypto Agility is the Key to Securing Internet of Things Identities
In this Spotlight Edition of the Security Ledger Podcast: identity is at the root of many of the security problems facing the Internet of Things, from vulnerable and “chatty” endpoints to a lack of robust update and lifecycle management features. To figure out how we might start to build a more secure IoT ecosystem, we invited Judah Aspler, the Vice President of IoT Strategy at Certified Security Solutions, or CSS Security in to talk about how more agile PKI infrastructure is one element in scaling the Internet of Things without creating a giant security mess.
FDA Medical Device Plan: a Baby Step in the Right Direction
In this industry perspective, Dan Lyon and Taylor Armerding of the firm Synopsys discuss the impact of the FDA’s new Medical Device Safety Action Plan, which promises to improve the cyber security of medical devices…eventually.
Spotlight: Deepika Chauhan of Digicert on the Challenges of Securing the Internet of Things
There’s an epidemic of insecure Internet of Things devices. But why? And what is the shortest path to ending that epidemic? In this Spotlight Edition* of The Security Ledger Podcast, we speak with Deepika Chauhan, the Executive Vice President of Emerging Markets at DigiCert. Her job: forging new paths for the use of public key encryption to secure Internet of Things ecosystems.